Our Vision
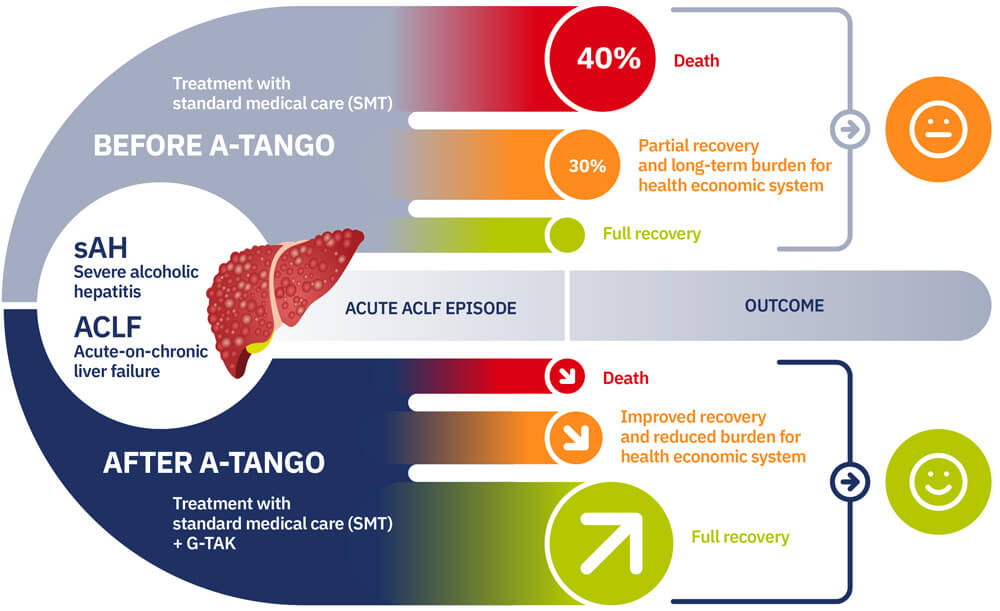
Various liver diseases can lead to severe scarring of the liver (cirrhosis), including autoimmune disorders, fatty liver, and bacterial or viral infections. Alcohol abuse, however, accounts for the majority (30-40%) of all liver-related deaths in Europe. A severe form of alcoholic liver disease is called steroid-resistant alcoholic hepatitis (sAH), which carries especially high mortality rates. Regardless of the cause, when cirrhosis progresses too far, the body can no longer compensate for the dysfunctional liver. This results, as the name implies, in so-called decompensated cirrhosis. More than 10 million people worldwide suffer from decompensated cirrhosis. In its final stages, decompensated cirrhosis leads to acute-on-chronic liver failure (ACLF), a syndrome characterised by systemic inflammation, fluid accumulation in the abdomen (ascites), impaired brain function (hepatic encephalopathy), internal bleeding (gastro-intestinal haemorrhage), and multi-organ failure, often leaving liver transplantation as the last resort to save the patient’s life. Therefore, effective treatment of ACLF remains an urgent and unmet need.
The A-TANGO consortium performs Phase 2 clinical studies of an innovative therapeutic strategy that improves hepatocyte proliferation through granulocyte colony-stimulating factor (G-CSF) and reduces systemic inflammation by repurposing TAK-242, an antagonist of toll-like receptor 4. We call this novel combinatorial therapy “G-TAK”. In addition, A-TANGO strives to identify reliable biomarkers for better patient stratification and an increased survival rate. Up to 25 liver centers in Europe will participate in the clinical study of A-TANGO.
Our vision is to drastically reduce the mortality rate of end-stage liver disease within the 5 years of the project and thereafter and to improve the prognosis, life expectancy, and quality of life of patients who suffer from decompensated cirrhosis.