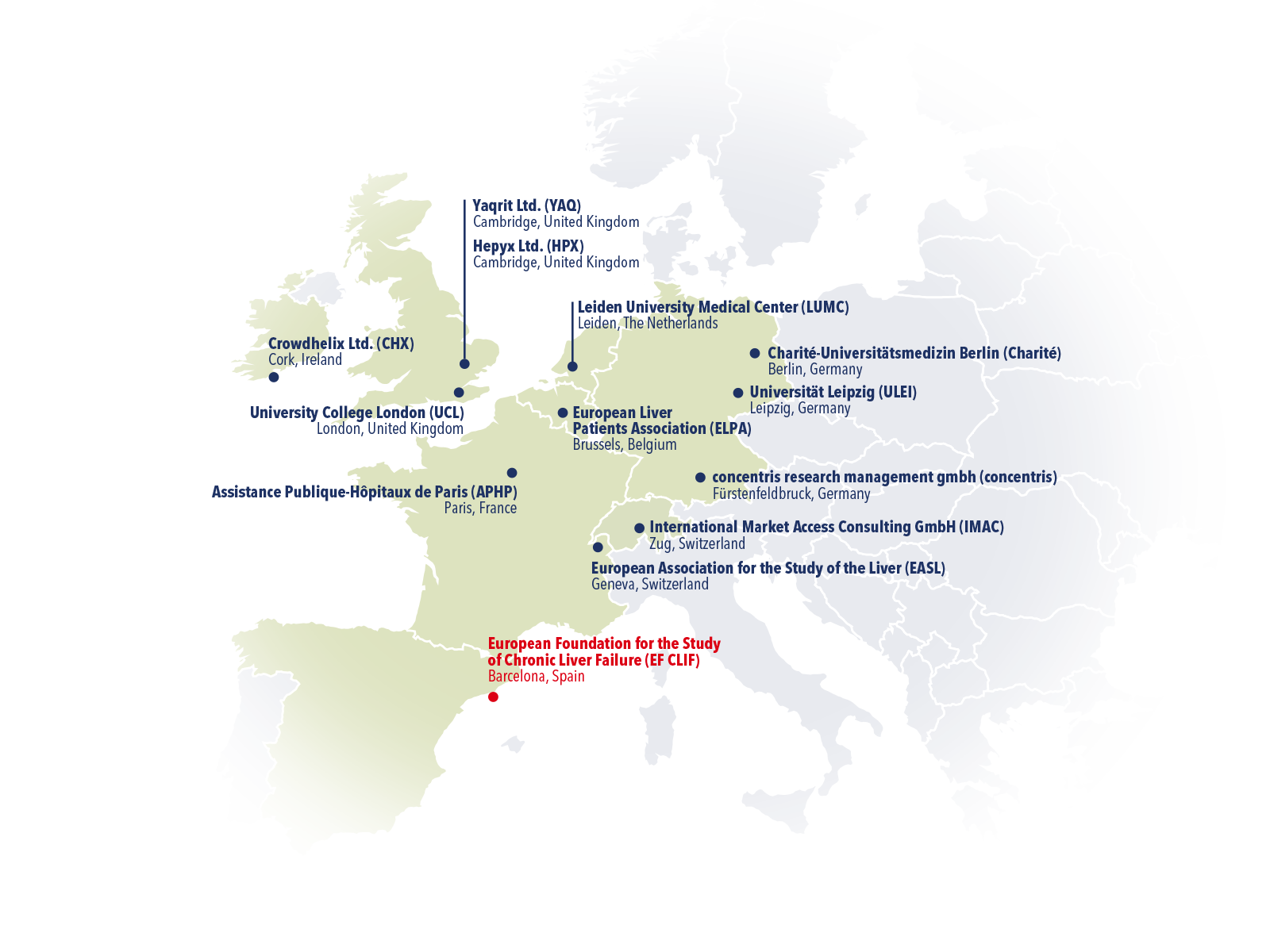
Members
The 13 institutions that collaborate in this multi-centre project are spread throughout Europe, and include world-class experts on liver cirrhosis, clinical trial management, drug manufacturing, molecular biology, data analysis, health economics, networking and dissemination. Our multidisciplinary team will ensure high-impact dissemination of scientific results and prepare an exploitation plan to raise commercial interest and facilitate smooth entry into clinical practice if G-TAK proves to be successful. The 13 institutions below are A-TANGO members and constitute the research consortium itself, while up to 25 hospitals in Europe will participate in the clinical trial. Soon, we will add a Study Sites page, listing all associated liver centers.